Cell migration is a widely researched and clinically relevant process that, with a greater understanding, may allow us to control a number of pathologies - arguably the most significant being cancer metastasis. However, cell motility has primarily been investigated using cell culture models, which involves watching cells move on 2-dimensional substrates. While these in vitro assays have been useful, there will always be questions surrounding the physiological relevance of studying cell movement in artificial conditions.
Drosophila macrophages (also known as hemocytes) can be live imaged during their embryonic dispersal which allows us to begin extrapolating our understanding of cell motility to cells within a living organism. This movie shows the migration of hemocytes as they disperse within a thin space beneath the epithelium and eventually adopt an evenly spaced pattern.
Drosophila macrophages (also known as hemocytes) can be live imaged during their embryonic dispersal which allows us to begin extrapolating our understanding of cell motility to cells within a living organism. This movie shows the migration of hemocytes as they disperse within a thin space beneath the epithelium and eventually adopt an evenly spaced pattern.
|
|
Once hemocytes have spread throughout the embryo, they do not stop migrating. They constantly patrol all of the surfaces within the embryo while maintaining an even cellular spacing. These cells are essential for a number of aspects of embryonic development, and we hypothesise that their constant movement and even distribution is critical for their functions. This movie shows hemocytes patrolling territory within the embryo while maintaining their even dispersal pattern.
|
|
|
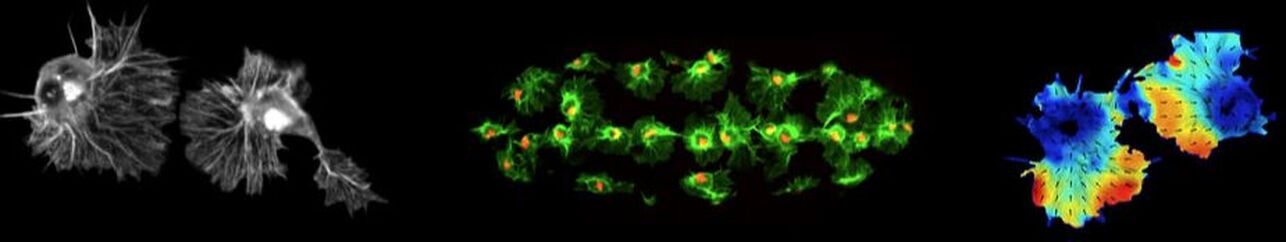
One of the interests of the laboratory is to understand how hemocytes maintain their even spacing within the embryo. We have shown that during their migration, hemocytes are undergoing contact inhibition of locomotion. This process of collision and subsequent repulsion is an instructive migratory cue that allows the cells to maintain space within the embryo. We are now investigating the cytoskeletal machinery and cellular recognition mechanisms that allow hemocytes to undergo this process with such reproducibility. This movie shows an example collision between two colliding hemocytes in which their actin and microtubule cytoskeletal networks are labeled.
|
|
|
Hemocytes can be imaged within the living Drosophila embryo at a resolution approaching what can be obtained from cells in culture. This has opened up the possibility of more detailed image analysis techniques that allow for subcellular quantification of protein dynamics within these cells. This movie shows colliding hemocytes in which their actin cytoskeleton was fluorescently labelled. Subsequently, the dynamics of the flowing actin networks was quantified by particle image velocimetry, which allows us to accurately measure the direction and speed of actin motion within these cells. Such computer vision techniques will allow us to precisely determine the intracellular mechanisms driving hemocyte movement.
|
|
|
|
Work in the laboratory is also revealing the function of hemocyte migration for Drosophila development. Recent work has shown that the reason for hemocyte dispersal is to allow these cells to evenly distribute extracellular matrix in the embryo. This movie shows embryonic hemocytes depositing Collagen IV, one of the major extracellular matrix components of the basement membrane. This is a surprising function for these cells, as extracellular matrix production is not thought to be a major function for macrophage populations. We are now using this model to understand how the basement membrane is constructed de novo during development and examine the function of this extracellular matrix for tissue morphogenesis.
|